These challenges are further compounded by ongoing business challenges especially in the realm of intellectual property (IP); where the so called “patent cliff” is an especially acute challenge for the pharmaceutical industry. Drug patents typically last for 20 years from the date of filing. However in purely commercial terms, a drug’s patent protection often amounts to less than 12 years since clinical trials and testing can last up to 8 years or longer.
For those unfamiliar, a “patent cliff” is a term of art describing the situation when a drug’s patent protection expires and denies the drug developer exclusivity on the manufacture of the drug that they developed – combined with the resulting loss of the opportunity to further recoup research and development costs. This allows for generic drug makers to manufacture budget alternatives at a much lower cost with the original drug developer and manufacturer losing as much as 90% of their original sales in the process. This situation was especially acute between 2007 and 2012 where over $60 billion in annual sales fell off a veritable “cliff” for many major drug companies in the United States.
While the expiration of patents is not unique to the pharmaceutical industry, its prevalence certainly is, which only serves to add to pharma’s many challenges which continue right up to the present.
In searching for greater commercial and processing efficiencies, it is surely time to reconsider the role of electrical tomography in the pharmaceutical industry and to reexamine some of Industrial Tomography Systems’ (ITS) previous collaborations with some of the world’s leading drug companies in order to inspire the scaled-up industrial application of a range of highly successful bench-top and pilot plant applications that have already been validated. This is even more pertinent given the exponential improvements in the data acquisition speeds of Industrial Tomography Systems’ (ITS) latest technology.
It was no surprise that electrical resistance tomography (ERT) should have found its way into the pharmaceutical industry with its powers of real-time visualisation of fluidised compounds and multiphase flows. “Monitoring of Multiphase Pharmaceutical Processes Using Electrical Resistance Tomography” is a seminal paper from 2005 which helped to identify ERT’s enormous pharmaceutical potential. Documenting a joint study between GlaxoSmithKline (GSK) and Imperial College London, the paper looked at ERT’s ability to monitor liquid-liquid dispersion and solid suspension in the production of the active ingredients that are foundational in many pharmaceutical products. The paper concluded that: “ERT is a valuable tool for the development of robust active pharmaceutical ingredient (API) manufacturing processes.”
In 2009, Industrial Tomography Systems (ITS) collaborated with Pfizer in a range of ERT applications across a number of Pfizer’s R&D and manufacturing centres in the United States including Groton, Connecticut and Kalamazoo, Michigan. Not surprisingly mix monitoring was the dominant application alongside monitoring paracetamol crystallisation. A summary of the 2009 collaboration between Industrial Tomography Systems (ITS) and Pfizer is illustrated in the table below.
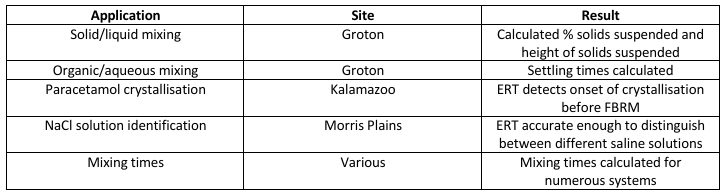
The conclusions were as compelling as they are clear. ERT was able to successfully monitor solid-liquid mixing, liquid-liquid mixing, and settling processes. While these applications ratified, and replicated, many existing and subsequent studies in the peer reviewed and grey scientific literature; the results were still successfully repeatable and worked perfectly with Pfizer’s own specific protocols and operational conditions.
Of particular note is the use of ERT to monitor paracetamol crystallization which replicated a paper quoted earlier in this article. The Pfizer collaboration used ERT to compliment focused beam reflectance measurement (FBRM) which was the monitoring technology already in place. While FBRM is a well-established technique to monitor fine particles, ERT showed its value by being able to visualise spatial inhomogeneity and void detection which enhances product quality.

In subsequent years, ERT has been shown to be very adept at monitoring crystallisation in a number of different pharmaceutical contexts. An excellent study published in 2022 demonstrated ERT’s proficiency in monitoring sucrose and calcium carbonate crystallization.
ERT’s repertoire of pharmaceutical applications has continued to grow as seen in a recent paper published in 2025. In this study ERT was used to monitor solid suspension and homogeneity of solids in relation to vaccine production. While aluminium hydroxide is used as an immunologic adjuvant in vaccine production, this study used hydrous magnesium silicate, for the purposes of the experiment, as it closely mimics the behaviour of aluminium hydroxide. The success of ERT was most compelling with the authors concluding:
“These findings confirm the viability of ERT for real-time suspension monitoring, demonstrating its potential for detecting solid accumulation and optimizing mixing in industrial and pharmaceutical processes, including vaccine manufacturing.”
The pharmaceutical industry’s “patent cliff” of the 2000s is likely to pale into insignificance compared with what is about to hit the industry by 2030. In 2023, PharmaVoice reported that 190 drugs were going to lose patent exclusivity by 2030 leading to revenue declines of 46% for the world’s ten biggest pharmaceutical companies.
Time and again, ERT has demonstrated its stellar credentials as a cost effective, non-invasive instantaneous monitoring technology whose real-time dynamic imagining can provide the pharmaceutical industry with the means to achieve major cost savings and process efficiencies across a wide range of mixing, crystallisation, and chromatography monitoring applications.
While it would be too bold a claim to assert that Industrial Tomography Systems’ (ITS) technology can be a panacea for the pharmaceutical industry’s impending “patent cliff”, electrical tomography can certainly provide a parachute.